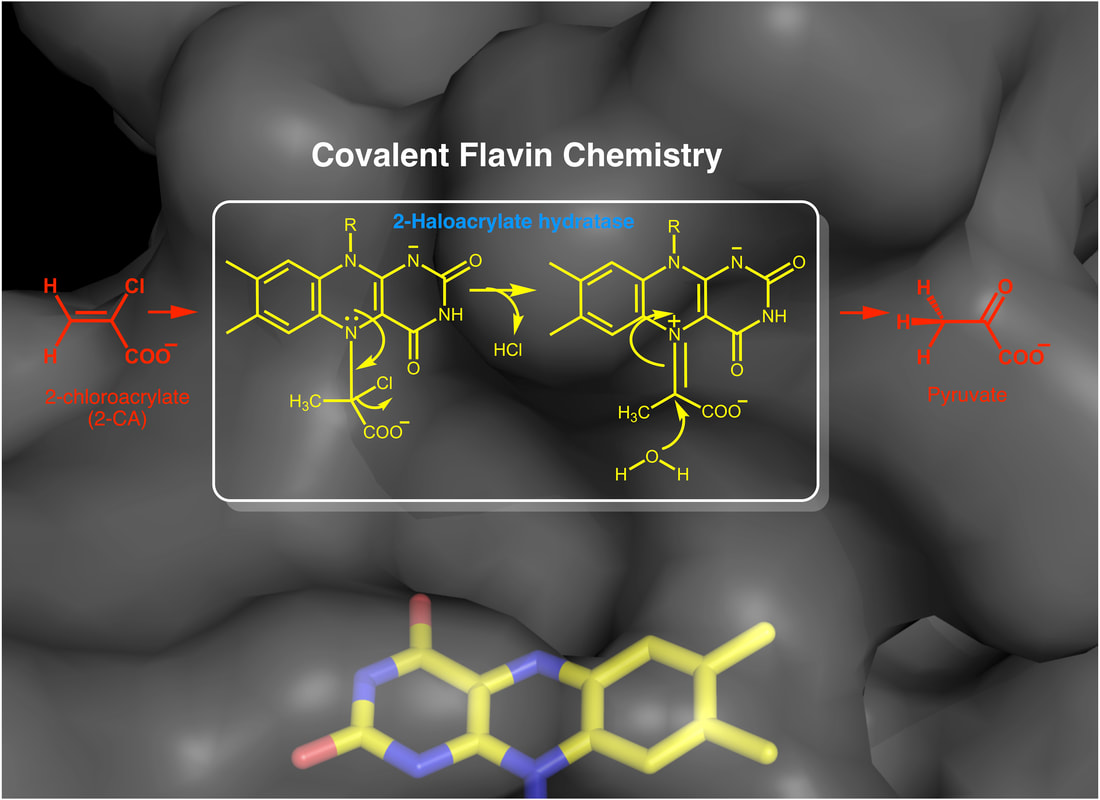
Non-redox dehalogenation reaction
Enzymes that contain flavin cofactor in the form of FMN or FAD are known as flavoenzymes. In this group of enzymes the flavin plays essential roles in biochemistry. In general, flavoenzymes utilize the redox properties of the isoalloxazine ring of the flavins to catalyze redox reactions. These reactions include electron transfer, dehydrogenations, hydroxylations, light and redox sensing, and epoxidations. Recently, new functionalities have been demonstrated for several flavoenzymes. These include the role of the flavin cofactor as an acid or base, as a nucleophile or electrophile, and in the formation of covalent intermediates, which can be considered noncanonical reactions that are catalyzed by atypical flavoenzymes. One focus of the project is to elucidate the protein-flavin interactions that tune the flavin for novel reactivity. The second is the mechanistic and structural characterization of 2-haloacrylate hydratase, an enzyme involved in bioremediation of halogenated compounds. Preliminary data strongly suggest that the mechanism of 2-HAH is novel