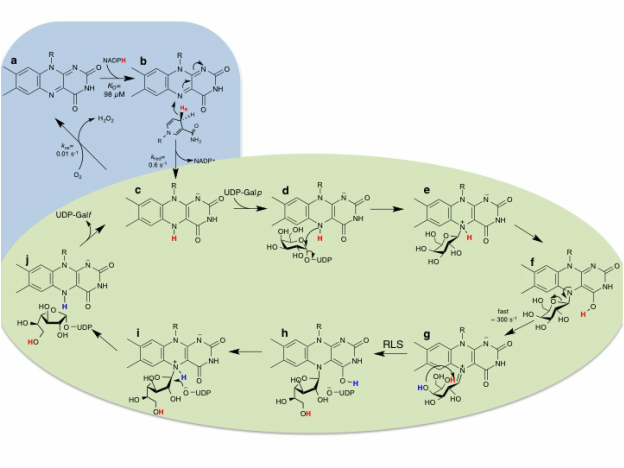
Eukaryotic UDP-galactopyranose mutases
Galactofuranose (Galf) is an important component of the cell wall of bacteria and fungi and is a virulence factor in protozoan parasites. Inhibition of UDP-galactopyranose mutase (UGM), the flavoenzyme that catalyzes the conversion of UDP-galactopyranose (UDP-Galp) to UDP-Galf, prevents growth of Mycobacterium, and deletion of the UGM gene dramatically attenuates virulence in Aspergillus and Leishmania species. Since UGM is not found in humans, inhibition of this enzyme is an attractive strategy for the development of antimicrobial agents.
In addition to its importance as a drug-design target, UGM is also a unique flavoenzyme that catalyses a net non-redox change in the formation of product. Although it is known that the reduced FAD is required for catalysis. Our group has been working on the eludation of the role of the reduced flaivn and the structural changes important for catalysis UGMs using enzymes from the fungus Aspergillus fumigatus (AfUGM) and the parasite Trypanosoma cruzi (TcUGM) as the prototypes.
Our biochemical, structural, and computational studies have shown that the flavin function as a nucleophile and proton shuttle mediated by bending of the flavin ring. Protein conformational changes mediate access to the active site by motion of the active site flaps. Bending of the flavin ring is facilitated by conformational change of the histidine loop. These data is driving our high throughput screening assays and computational drug screening and design.
In addition to its importance as a drug-design target, UGM is also a unique flavoenzyme that catalyses a net non-redox change in the formation of product. Although it is known that the reduced FAD is required for catalysis. Our group has been working on the eludation of the role of the reduced flaivn and the structural changes important for catalysis UGMs using enzymes from the fungus Aspergillus fumigatus (AfUGM) and the parasite Trypanosoma cruzi (TcUGM) as the prototypes.
Our biochemical, structural, and computational studies have shown that the flavin function as a nucleophile and proton shuttle mediated by bending of the flavin ring. Protein conformational changes mediate access to the active site by motion of the active site flaps. Bending of the flavin ring is facilitated by conformational change of the histidine loop. These data is driving our high throughput screening assays and computational drug screening and design.