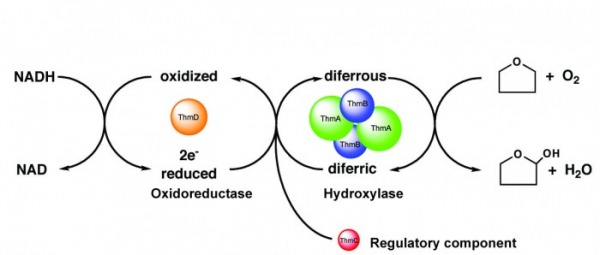
Tetrahydrofuran monooxygenase
Tetrahydrofuran monooxygenase (Thm) is one of the two members of Group 5 baterial multicomponent monooxygenases. Thm was identified from Pseudonocardia sp. strain K1, which is capable of growing aerobically on tetrahydrofuran as the sole carbon source. The hydroxylase enzyme of Thm is composed of only two subunits, which in solution are predicted to form a heterotetramer. In all of the BMM enzymes studied to date, the hydroxylase is composed of a dimer of three polypeptide chains. Therefore, this novel member of the BMM family represents a “simplified” monooxygenase complex. The oxidoreductase component, ThmD, is composed of an NADH-and FAD-binding domain and a [2Fe-2S] domain. ThmD is responsible for transferring electrons from NADH to the diiron center via the FAD and the [2Fe-2S] center. A unique feature of ThmD is that the flavin is covalently attached. Our research focuses on the reconstitution of the activity of Thm in vitro. In particular, we are interested in determining the nature of the covalent flavin attachement in ThmD.