N-Monooxygenases (NMOs)
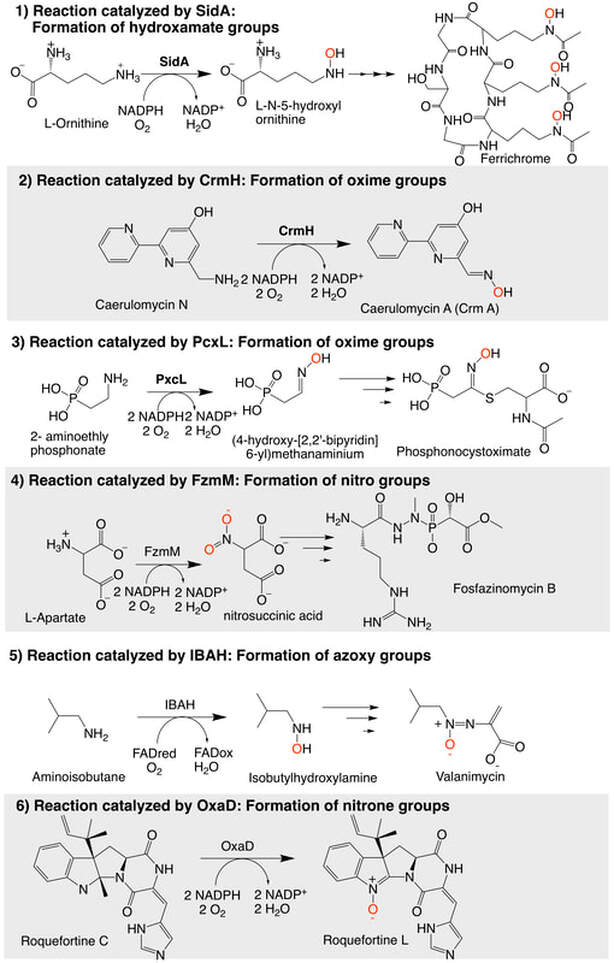
We are working with the growing family of flavin-dependent enzymes that hydroxylate nitrogen atoms: N-monooxygenases (NMOs). We are focusing on enzymes involved in pathogenesis or the biosynthesis of natural products with pharmaceutical activities. We hope to use mechanistic and structural biochemistry to understand in detail the mechanism of action with the hope of to identify inhibitors or use these enzymes in the production of novel antibiotics. For the past 10 years, we focused on enzymes involved in siderophore biosynthesis (SidA, MbtG). We have recently selected uncharacterized NMOs that are involved in the formation of oxime functional groups in phosphonocystoximate (i.e., PcxL) and caerulomycin A (i.e., CrmH) biosynthesis, nitro groups in fosfazinomycin biosynthesis (i.e., FzmM), azoxy groups in valanimycin biosynthesis (i.e., IBAH), and nitrone containing compounds (i.e., OxaD).